In Stock CAS 38304-91-5 Anti Hair Loss Raw Material Pure Bulk Powder 99% Minoxidil With Fast Delivery
Product Introduction
Minoxidil was the first drug approved by the FDA for the treatment of androgenetic alopecia (hair loss). Before that, minoxidil had been used as vasodilator drug prescribed as oral tablet to treat high blood pressure, with side effects that included hair growth and reversal of male baldness. In the 1980s, UpJohn Corporation came out with a topical solution of 2% minoxidil, called Rogaine, for the specific treatment of androgenetic alopecia. Since the 1990s, numerous generic forms of minoxidil have become available to treat hair loss while the oral form is still used to treat high blood pressure.
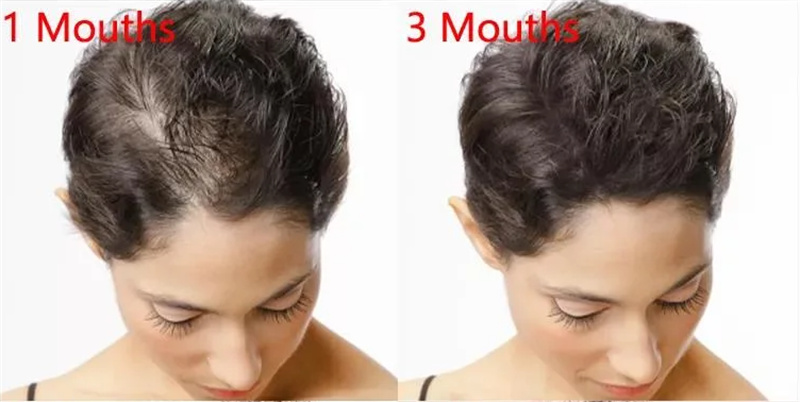
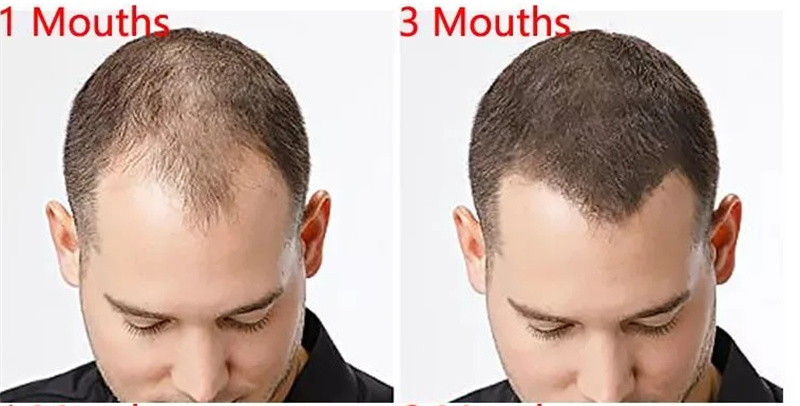
Minoxidil is a vasodilator medication known for its ability to slow or stop hair loss and promote hair regrowth. It is available over the counter for treatment of androgenic alopecia, among other baldness treatments, but measurable changes disappear within months after discontinuation of treatment. Its effectiveness has largely been demonstrated in younger men (18 to 41 years of age), the younger the better, and in those with balding in the central (vertex) portion of the scalp.
Function
1. Promote hair regrowth.
2. Slow or stop hair loss.
3. Treat high blood pressure.
Action Mechanism
The mechanism by which minoxidil promotes hair growth is not fully understood. The drug has a nitric oxide group in its structural formula and may act as a nitric oxide dosing agent. It is also a potassium channel opener, causing overpolarization of the cell membrane.For extensive hair loss, minoxidil is less effective.In addition, for young men with less than five years of hair loss problems, it has a very significant effect.Currently, minoxidil is only used for hair loss on the top of the head. It is also a vasodilator.In theory, by dilating blood vessels and opening potassium channels to bring more oxygen and nutrients to the hair follicles, the follicles would move from resting to growing.
What Can We Do?
1. Free samples can be provided upon your request.
2. Production capacity: 20 tons/month.
3. The factory covers an area of 7,000 square meters and has 4 Ph.D. technical engineer.
4. Transportation method: express, air transport, sea transport
5. Quality control: 3rd party laboratory testing by Eurofins, SGS, BV etc.
6. 24 hours and 7 days full time standing-by services.
Product Data Sheets
| Analysis | Description | Test Method |
| Appearance | White Powder | Visual |
| Odor | Characteristic | Nasil |
| Identification | Conform with reference sample | HPLC |
| Mesh Size | 100% pass 80 mesh | CP2020 |
| Moisture Content | ≤ 0.5% | GB5009.3-2016 |
| Residue on Ignition | ≤ 0.5% | GB5009.4-2016 |
| Heavy Metals | ≤ 10 ppm | CP2020 |
| Arsenic (As) | ≤ 2.0 ppm | BS EN ISO17294-2 2016 2016 |
| Lead (Pb) | ≤ 3.0 ppm | BS EN ISO17294-2 2016 |
| Cadmium (Cd) | ≤ 1.0ppm | BS EN ISO17294-2 2016 |
| Mercury (Hg) | ≤ 0.1ppm | BS EN ISO17294-2 2016 |
| Total Plate Count | ≤ 1000 cfu/g | ISO 4833- 1:2013 |
| Yeast and Mold | ≤ 100 cfu/g | ISO 21527-2:2008 |
| Escherichia Coli | Absent | ISO 16649-2:2001 |
| Salmonella/25g | Absent | ISO 6579-1:2017 |
| Assay | ≥99% | HPLC |
Packing & Shipping

What We Can Do?